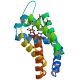
Mapping the catalytic cycle of Schistosoma mansoni thioredoxin glutathione reductase by X-ray crystallography
Angelucci F, Dimastrogiovanni D, Boumis G, Brunori M, Miele AE, Saccoccia F, Bellelli A.
Schistosomiasis is the second most widespread human parasitic disease. It is principally treated with one drug, praziquantel, that is administered to 100 million people each year; less sensitive strains of schistosomes are emerging. One of the most appealing drug targets against schistosomiasis is thioredoxin glutathione reductase (TGR).
This natural chimeric enzyme is a peculiar fusion of a glutaredoxin domain with a thioredoxin selenocysteine (U)-containing reductase domain.
Selenocysteine is located on a flexible C-terminal arm that is usually disordered in the available structures of the protein and is essential for the full catalytic activity of TGR.
In this study, we dissect the catalytic cycle of Schistosoma mansoni TGR by structural and functional analysis of the U597C mutant. The crystallographic data presented herein include the following: the oxidized form (at 1.9 Å resolution); the NADPH- and GSH-bound forms (2.3 and 1.9 Å, respectively); and a different crystal form of the (partially) reduced enzyme (3.1 Å), showing the physiological dimer and the entire C terminus of one subunit.
Whenever possible, we determined the rate constants for the interconversion between the different oxidation states of TGR by kinetic methods. By combining the crystallographic analysis with computer modeling, we were able to throw further light on the mechanism of action of S. mansoni TGR. In particular, we hereby propose the putative functionally relevant conformational change of the C terminus after the transfer of reducing equivalents from NADPH to the redox sites of the enzyme.