 The structure of carbonmonoxy neuroglobin reveals a heme-sliding mechanism for control of ligand affinity.
The structure of carbonmonoxy neuroglobin reveals a heme-sliding mechanism for control of ligand affinity.
Vallone, B., Nienhaus, K., Matthes, A., Brunori, M., Nienhaus, G.U.
Neuroglobin (Ngb), a globular heme protein expressed in the brain of vertebrates, binds oxygen reversibly, with an affinity comparable to myoglobin (Mb). Despite low sequence identity, the overall 3D fold of Ngb and Mb is very similar.
Unlike in Mb, in Ngb the sixth coordination position of the heme iron is occupied by the distal histidine, in the absence of an exogenous ligand. Endogenous ligation has been proposed as a unique mechanism for affinity regulation and ligand discrimination in heme proteins. This peculiarity might be related to the still-unknown physiological function of Ngb.
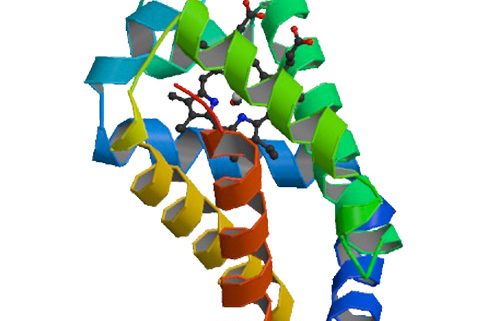
Here, we present the x-ray structure of CO-bound ferrous murine Ngb at 1.7 A and a comparison with the 1.5-A structure of ferric bis-histidine Ngb.
We have also used Fourier transform IR spectroscopy of WT and mutant CO-ligated Ngb to examine structural heterogeneity in the active site.
Upon CO binding, the distal histidine retains (by and large) its position, whereas the heme group slides deeper into a preformed crevice, thereby reshaping the large cavity ( approximately 290 A(3)) connecting the distal and proximal heme sides with the bulk. The heme relocation is accompanied by a significant decrease of structural disorder, especially of the EF loop, which may be the signal whereby Ngb communicates hypoxic conditions.
This unexpected structural change unveils a heme-sliding mechanism of affinity control that may be of significance to understanding Ngb’s role in the pathophysiology of the brain.
Related Structures:
Primary Citation of: 2W92, 1Q1F, 2VRY